What You Should Know About Organic Light-Emitting Diode (OLED) Technology
A thorough explanation of OLED technology and its applications. Like an LED, an OLED is a solid semiconductor device that is 100 to 500 nanometers in length or 200 times smaller than a human hair!
A thorough explanation of OLED technology and its applications. Like an LED, an OLED is a solid semiconductor device that is 100 to 500 nanometers in length or 200 times smaller than a human hair!
An organic light-emitting diode (OLED) is a solid state device similar to the light-emitting diode (LED) but composed of several layers of material. An OLED is flat light emitting technology composed by placing a series of extremely thin organic films between two conductors. When a potential difference occurs between the conductors, a bright light is emitted. This technology can be used to produce curved display panels, smart watch faces, and televisions. Since OLEDs emit light, the panels do not require a backlight like liquid crystal display (LCD) televisions do, making them thinner and more efficient.
Components of an OLED
OLEDs can be composed of either two or three layers of organic material; a third layer helps transfer electrons from the cathode to the emissive layer more easily. Focusing on the two layer design, an OLED consists of the following components:
- Substrate - This is the base that supports the OLED. It can either be made of a thin translucent glass or foil material.
- Anode - When a potential difference occurs, the anode loses electrons (or "receives holes"). The anode is also known as the emitter.
- Organic layer - The layers that sit on top of the anode are made of organic molecules, such as hydrogen or carbon, that contain bonds or conductive polymers.
- Conductive layer - Composed of organic plastic molecules that are used to transport "holes" from the anode.
- Emissive layer - This layer is composed of organic materials as well, but different than those used in the conducting layers. This helps transfer electrons from the cathode and emits light.
- Cathode - The cathode injects electrons when an electrical current flows through the OLED. Such metals as aluminum and calcium are used in the cathode and it may be transparent or not. The cathode is also known as the conductor.

Emitting Light
Similarly to LEDs, OLEDs emit light through a process called electro-phosphorescence. This process is a form of phosphorescence that emits light through a substance without combustion or perceptible heat. The process can be broken down into six steps:
- The power supply of the device will apply a potential difference across the OLED, from the cathode to the anode.
- Now there is an electrical current that flows from the cathode (negative terminal of the diode) to the anode (positive terminal of the diode) through the organic conductive layers. The cathode is now able to give electrons to the emissive layer of the organic molecules. After the electrons pass through the emissive layer, they are transferred to the conductive layer of organic molecules. At this point, the anode now removes electrons passed through from the cathode. This separation of electrons is equivalent to giving electron holes to the conductive layer.
- Electrons will find these electron holes between the conductive and emissive layers of organic molecules. As an electron fills a hole, the electron is forced to give up energy. The energy required for electrons to fill an electron hole is in the form of photons of light.
- As an electron fills an electron hole, the energy is released and light is emitted.
- The color of light that the electrons emit depends on the type of organic molecules in the emissive layer. To produce different colors, manufacturers place several different types of organic films on one single LED to make various color arrays.
- The amount of voltage applied from the power supply will affect the intensity or brightness of the light emitted from the electrons. The higher the current flowing from cathode to anode, the brighter the light.
AMOLED vs PMOLED
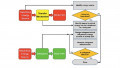
AMOLED and PMOLED use two different drivers in OLED displays. A PMOLED (Passive-Matrix OLED) is restricted to its size and resolution, but is much cheaper and easier to manufacture than an AMOLED (which uses an Active-Matrix). An AMOLED utilizes an active-matrix TFT (thin filter transistor) array and storage capacitors. While AMOLED displays are much more efficient and can be made larger than normal, they are much more complicated to manufacture. AMOLEDs are best used in computer monitors, large-screen televisions, and electronic billboards. You'll find PMOLEDs in devices such as cell phones, PDAs, and MP3 players.
In AMOLEDs, the active-matrix describes how each OLED is controlled. Using an m x n matrix, the number of connectors needed to control the display is m + n. An individual pixel is attached to a switch device, which actively maintains the pixel state while other pixels are being controlled. This control scheme prevents state changes in one pixel from interfering with the states of other pixels.
PMOLEDs have strips of cathodes, organic layers, and anodes. The anode strips are laid perpendicular to the cathode strips. Each intersection of where a cathode and anode strip meet, make up a pixel, where light is emitted. This design uses a simple control scheme in which each row in the display is controlled sequentially. This is a matrix scheme meaning that only m + n control signals are require to address an m x n display. A pixel in this passive-matrix must maintain its state without active driving circuitry until it can be refreshed again. However, unlike AMOLED, PMOLED electronics do not contain a storage capacitor. The figure below is a visual guide between the design and fabrication of AMOLEDs and PMOLEDS.

With an almost instantaneous refresh rate, OLED displays do not suffer from motion lag or motion blur as do LED displays. Since OLEDs generate their own light, unlike LCDs, they do not need a backlight and therefore have deeper blacks, brighter whites and gray scales in between can be easily achieved. Both active and passive matrix structures operate each pixel individually, offering maximum efficiency and the greatest contrast ratio. While the light-emitting layers of an OLED are much thinner and lighter, the substrate of an OLED can be flexible instead of a standard rigid LED. Due to OLEDs being much more efficient than LCDs, there is no doubt that their presence in the market will increase.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin










This is fascinating in its own right with the potential for flexible screens, lighting etc. However, the notion occurs to me that this could lead to a potential revolutionary breakthrough in solar cell technology - since the process described here is essentially the reverse of how solar panels generate potential difference and current. If light could be captured by these or similar devices and the process reversed, then a new generation of flexible solar cells made from cheap, readily available organic materials would begin, revolutionizing the industry and the world.