Ingestible Sensor Completes First In-Human Trial to Monitor Vital Signs
MIT has reported the first-ever in-human trial of an ingestible vitals-monitoring pill.
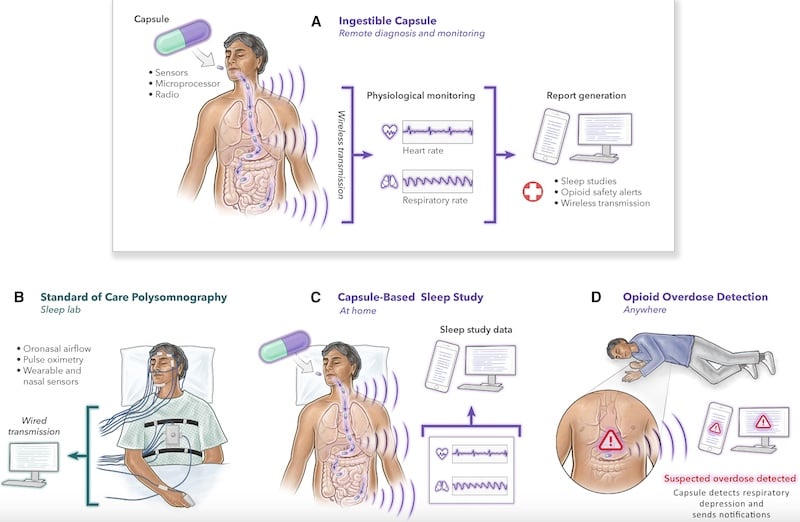
MIT recently published a study on an ingestible sensor designed to monitor vital signs like heart and breathing rates. The device, encapsulated in a pill no larger than a multivitamin, has shown promise in diagnosing sleep apnea and identifying opioid overdose risks.
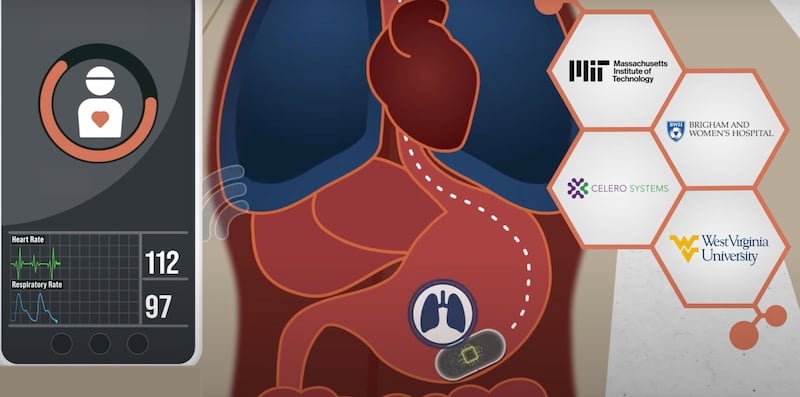
MIT ingestible sensor. Screenshot (modified) used courtesy of MIT
How does this sensor monitor these biometrics unobtrusively while withstanding the conditions of the human body?
A Hardware Breakdown of MIT’s Ingestible Sensor
The Vital Monitoring (VM) pill employs a range of off-the-shelf integrated circuits and electronic sensors. The core components include the following:
- Microcontroller: STM32L4, an Arm Cortex M4-based, 32-bit MCU (STMicroelectronics)
- Accelerometer: ADXL363, a three-sensor combination including acceleration and temperature (Analog Devices)
- Memory: CY15B108QI, an 8-Mbit FRAM (Infineon)
- Radio: ZL70323MNJ, a MICS 400-MHz module (Microchip)
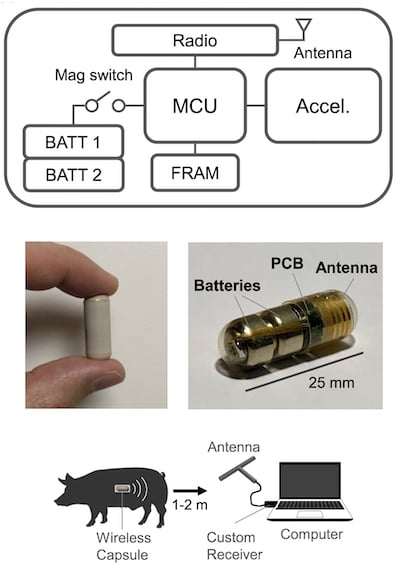
A high-level schematic of the VM pill electronics along with its experimental setup for porcine testing. Image (modified) used courtesy of Science Direct
Powering the device are two silver oxide button cell batteries. The pill also incorporates two custom-printed circuit boards and a magnet-sensing circuit for power control. The device communicates wirelessly with a custom-built receiver connected to a laptop, which runs both data acquisition and analysis software.
The pill’s outer shell, measuring 9 mm in diameter and 25 mm in length, is crafted from biocompatible polyether ether-ketone plastic 0.5 mm thick.
The Electronic Pill Undergoes Animal and Human Testing
Porcine studies demonstrated the robustness of the VM pill, where it achieved a reliable wireless connection up to two meters from the animal's abdominal wall. The accelerometer data correlated closely with veterinary lab vitals monitoring equipment, with mean absolute errors of 0.5 breaths/min for respiratory rate (RR) and 1.6 beats/min for heart rate (HR).
MIT researchers achieved the first human trial of the ingestible sensor. Image used courtesy of Science Direct
In human trials involving 10 subjects, the VM pill accurately captured vital signs, displaying distinct respiratory and cardiac frequency bands. The data processing techniques precisely measured RR and HR, with gaps in data during wireless transfers and intervals of excessive movement or peristalsis. The VM pill accurately measured RR within one breath per minute compared to polysomnography (PSG) airflow data, and its HR accuracy was within two to three beats per minute compared to PSG photoplethysmograph data.
The VM pill's accuracy in measuring RR and HR is comparable to clinically used devices, indicating its potential as a viable alternative for monitoring vital signs. The researchers suggest the pill can be used to detect sleep apnea and opioid-induced respiratory depression (OIRD), which are physiologically similar.
Next Steps to Commercialization
MIT's VM pill represents a pioneering step in medical diagnostics and patient monitoring. It offers a non-invasive, efficient, and accurate method for vital signs monitoring, with potential applications in diagnosing conditions like sleep apnea and detecting OIRD.
Despite their clinical promise, ingestible sensors face several production challenges moving forward. Developers must first ensure complete biocompatibility and safety, particularly over extended periods. They must also prioritize data security and privacy, given the sensitive nature of the health data collected. Additionally, the device must maintain continuous and reliable data transmission from within the human body and support a long battery life.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin