Aluminum: Yet Another Potential Alternative to Lithium-Ion Batteries
While lithium reigns as the dominant material in battery technology, researchers are exploring a more abundant (and affordable) alternative—aluminum.
In fast-charging electric vehicles (EVs) and portable devices alike, battery research is an ever-evolving point of interest for electronic designers. While lithium- and lead-based batteries have dominated the market for a long time, these materials are prone to burnout and sourcing issues.
A newcomer has emerged as a potential material to be used in battery manufacturing, and it’s not silicon; it’s aluminum.
Why Aluminum?
Lithium, which is found in most batteries in some form, is broken down from hard rock and brine deposits and can be created by mixing a few chemicals and minerals. However, lithium's usefulness falls short when it lacks complementary materials like cobalt and phosphorous, which face sourcing and manufacturing challenges.
Meanwhile, aluminum is one of the most abundant metal materials on earth. In recent years, research suggests that aluminum-based batteries would be cost-efficient, lightweight, and easy to obtain compared to their lithium counterparts.

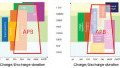
Comparison of aluminum to lithium. Image used courtesy of Frontiers in Chemistry
The problem is that aluminum is tricky to integrate into battery electrodes. Several research groups worldwide have sought ways to integrate aluminum in standard battery processing methods. From those studies, aluminum has shown promising takeaways that involve energy storage and solid recharging capabilities; for instance, aluminum has four times the volumetric capacity of lithium analog.
Cornell University's Aluminum Battery
Recently, researchers from Cornell University studied various low-cost materials to create a rechargeable battery that relies on aluminum-ion materials.
Within the battery itself, researchers observed a glass fiber separator that divided the anode and cathode portions of the battery. That glass fiber collided with the aluminum particles and ended up short-circuiting, which led to battery failure.

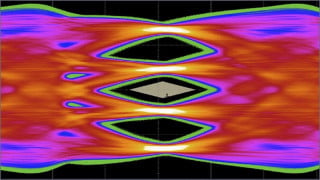
Aluminum deposited on carbon fibers inside the battery cell. Image used courtesy of Cornell University
The researchers solved this issue by designing a substrate of interwoven carbon fibers to form a stronger chemical bond with the aluminum material. As the battery charged, aluminum was deposited into the carbon structure and held together in a multi-layered substrate, avoiding contact with the glass fiber separator.
This study from Cornell suggests that aluminum-anode batteries can be reversibly charged and discharged several orders. From their study, the researchers discovered that zinc-anode aluminum batteries yielded a long cycle lifespan.
Aluminum Can Unlock Fast-charging Capabilities
On the other side of the world, China’s Dalian University of Technology (DUT) recently teamed up with the University of Nebraska (UNL) to develop a reliable, portable, fast-charging, aluminum-based solution.
The team of combined researchers found that in order to have a battery accept faster-charging abilities, the component would require a large current injection. However, introducing a larger current would result in a larger drop in overall resistance.
The DUT and UNL researchers added a three-dimensional network of graphene (acting in a similar fashion to Cornell’s carbon fibers), preventing the aluminum from bonding to the separator. The graphene helped boost the charge capacity of the battery. But once the graphene material grabbed ahold of anions that build up in the cell, it had difficulty releasing them.

3D graphene network after supercritical carbon dioxide drying. Image used courtesy of Nature
This meant that while graphene and aluminum are a good start for showcasing energy storage, they may not be efficient for dissipating charge to a device. To solve this issue, developers would need to deposit an extra amount of aluminum to balance out the graphene and allow for energy to dissipate easier.
The study claims that an aluminum-ion battery could charge a device within 0.35 seconds.
"Al-Air" and the Future of Aluminum Batteries
As the Cornell and DUT-UNL researchers suggest, the key to aluminum-based batteries may be in an interwoven, 3D-layer material coupled with aluminum. This, in turn, can create a battery cell that offers large energy storage and quick energy dissipation.
Some companies have already commercialized aluminum-based batteries. For instance, Israeli startup Phinergy and the Indian Oil Corporation have teamed up to create something called an "Aluminum-Air" (Al-Air) battery for EVs. This technology functions when the Al-Air battery draws in oxygen from its surrounding environment, which reacts with water and aluminum to create electricity. The battery can be reloaded with new anodes and electrolytes when the aluminum fully erodes.

Working principle of an aluminum-air battery. Image used courtesy of The Times of India
While aluminum-based batteries are still a long way from mainstream commercialization, it's possible that with continued research this material may one day stand as an alternative to standard lithium-ion solutions.
Potential Alternatives to the Lithium-Ion Battery
Aluminum is by no means the only material researchers are exploring as an alternative to lithium-based batteries. Check out some other materials of interest.
- Can Sodium-Ion Dethrone Lithium-Ion as a Dominant Battery Technology?
- General Motors Bets on Lithium Metal—Not Lithium Ion—as Path Forward for EV Batteries
- Are Lithium-Ion Batteries Here to Stay? The Promise of Li-Ion Batteries with Silicon Anodes

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin