Chinese Research Potentially Verifies Stanford University ‘Nickelate’ Superconductivity
Last year, scientists discovered a promising class of materials that could be used as superconductors—the so-called “Nickelates”.
There has been a huge amount of hype surrounding these materials, with some commentators going so far as to say they herald the start of the “Nickel Age” of superconductivity. This is because nickelates are able to conduct electrical current without any resistance, even at high temperatures.
It was soon discovered, as recently reported by AAC, by Austrian researchers that the initial results from Stanford, which reported these properties, could not be reproduced: This is because in some nickelates, additional hydrogen atoms are incorporated into the material’s structure, completely changing its electrical behavior.
Investigating the Electronic Structure of Nickelates
Now, a team at the Ningbo Institute of Materials Technology and Engineering (NIMTE) of the Chinese Academy of Sciences has conducted investigations into the electronic structure of the recently discovered nickelate superconductors.
Their work, which was published in Physical Review Letters on April 22, successfully explains the experimental difficulties that were experienced by the Vienna team when they tried to synthesize superconducting nickelates.
Accounting for Synthesizing Difficulties
To clarify the claims of the Vienna researchers and others that nickelates could not be reproduced and in fact did not exhibit superconducting properties at all, the NIMTE research team performed calculations based on density functional theory and dynamic mean-field theory by using supercomputers.
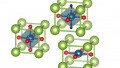
The team found that the chemical reduction of an ABO3-type compound with CaH2 may result in both ABO2 and ABO2H. It was also discovered that the topotactic hydrogen in nickelates was energetically favorable for LaNiO2 but not for strontium doped NdNiO2, leading to dramatic consequences for the electronic structure: That of 3d9 LaNiO2 is similar to (doped) cuprates, while 3d8 LaNiO2H is a two-orbital Mott insulator.
An illustration of hydrogen incorporated into the nickelate structure. Image credited to TU Wien
Difficulty in Replicating Results
Hydrogen can therefore be incorporated into the material structure of some nickelates which completely changes the material’s electronic properties. This proposal was verified by researchers from the National University of Singapore (NUS) who extracted the hydrogen that was released in the production process and succeeded in synthesizing superconducting nickelates.
The NIMTE team’s study, along with the NUS team’s findings and successful synthesizing of superconducting nickelates, may account for the Vienna team’s difficulties in achieving the same and also explain why some nickelates are superconducting while some others are not.
Additionally, the NIMTE team’s study suggests potential improvements on the original synthesizing process to improve success rates: Compressive strain and strontium doping, long reaction times to reduce hydrogen gas pressure, and low temperatures.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin